The particles which are structureless, invisible and not regarded as made up of some other particles are called elementary particles. Hundreds of the elementary particles are discovered so far. These particles are fundamental in the sense that these particles can not be explained as the system of other particles.
The idea that the world is made up of fundamental particles has a long history. In about 400 B.C., the Greek philosophers Democritus and Leucippus suggested that matter is made up of indivisible particles which they called atoms, which is derived from a – (not) and tomos (cut or divided). This idea remained dormant until about 1804, when John Dalton discovered that many chemical phenomena could be explained if atoms of each element are the basic, indivisible building blocks of matter.
Until the discovery of electron by J.J. Thomson in 1897, it was assumed that atoms were fundamental constituent of all the matter. The discovery of the idea of quanta, the bundles of electromagnetic radiation by Einstein in 1905 revolutionized the understanding of electromagnetic radiation and brought about quantum mechanics. From Thomson’s discovery of electron and Rutherford’s discovery of atomic nucleus and proton in 1911 made it apparent that atoms were not fundamental in the sense that they have an internal structure. The discovery of neutron by James Chadwick in 1932 completed a model of atom consisting protons and neutrons surrounded by sufficient electrons to balance the nuclear charge.
This model did not however explain the stability of nucleus. Yukawa in 1935 proposed a concept of exchange force which is responsible for the stability of nucleus. This involves exchange of shirt lived particles between the nucleons in the nucleus. These exchange particles are called mesons. This concept leads to the discovery of strong interaction and weak interaction bringing the total number of interactions to four. It also leads to the discovery of some 200 short lived elementary particles some of which are clearly more elementary than the others.
A brief description of some important particles is as follows:
- Electron: It is the first fundamental particle which was discovered by Thomson in 1897. It revolves around the nucleus of an atom in different orbits. Electron plays an important role in explaining the physical and chemical properties of substances. Its charge is -1.6 × 10-19C and its mass is 9.1 × 10-31kg. Its symbol is e or -1e0.
- Proton: It was discovered by Rutherford in 1911. It has a positive charge (+1.6 × 10-19C) equal to the electronic charge and its mass (1.626 × 10-37kg) is 1836 times the electronic mass. In free states, the proton is stable particle. Its symbol is p. It is also written as 1H1.
- Neutron: It was discovered by Chadwick in 1932. It carries no charge. Its mass (1.6749 × 10-27kg) is 1839 times the electronic mass. In free state the neutron is unstable (its mean life is about 17 minutes), but it constitutes a stable nucleus along with proton. Its symbol is n or on1.
- Positron: It was also discovered in 1932 by Anderson. Its charge and mass are same as those of electron, the only difference being that it is positively charged whereas the electron is negatively charged. Its symbol is e+ or +1e0.
- Antiproton: It was discovered in 1955. Its charge and mass are same as those of proton, the only difference being that it is negatively charged. Its symbol is p̅.
- Antineutron: It was discovered in 1956. It has no charge and its mass is equal to the mass of neutron. The only difference between neutron and antineutron is that if they spin in the same direction, their magnetic moments will be in opposite directions. The symbol for antineutron is n̅.
- Neutrino and Anti-neutrino: The existence of these particles was predicted in 1930 by Pauli while explaining the emission of β-particles from radioactive nuclei, but they were observed experimentally in 1956. Their rest-mass and charge are both zero but they have energy and momentum. Both neutrino and anti-neutrino are stable particles. The only difference between them is that their spins are in opposite directions. Their symbols are ν and ν̅ respectively.
- Pi-mesons: The existence of these particles was predicted by Yukawa in 1935 as the originator of exchange-forces between the nucleons, but they were actually discovered in 1947 in cosmic rays. Pi-mesons are of three types:
- Positive pi-meson: It is a positively charged particle whose charge is equal to the electronic charge and whose mass is 274 times the electronic mass. It is an unstable particle. Its mean life is of the order of 10-8 second. Its symbol is π+.
- Negative pi-meson: It is a negatively charged particle whose charge is equal to the electronic charge and whose mass is 274 times the electronic mass. Its mean life is also of the order of 10-8 second. Its symbol is π–.
- Neutral pi-meson: This particle has no charge. Its mass is nearly 264 times the electronic mass. Its mean life is of the order of 10-15 second. Its symbol is π0. On disintegration, it forms two γ-photons.
- Photons: These are the bundles of electromagnetic energy and they travel with the speed of light. If the frequency of waves be ν, then the energy of a photon is hν and momentum is hν/c. Its symbol is γ.
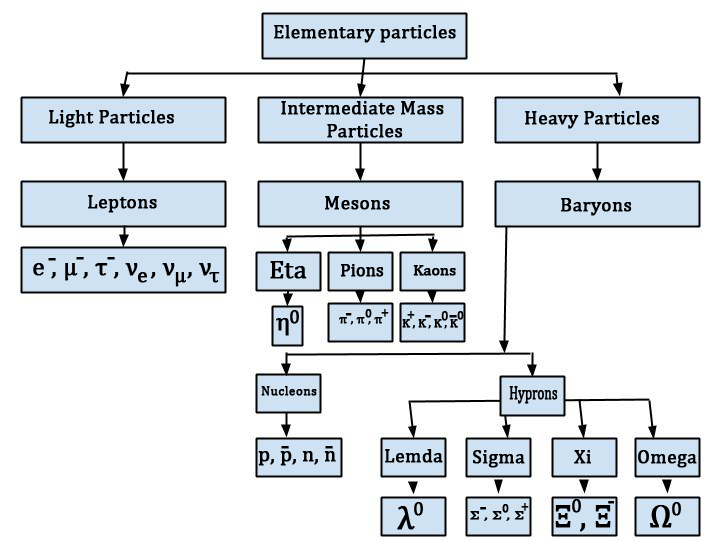
Classification of Elementary Particles
Characteristic Properties of Elementary Particles
The elementary particles are characterized by their mass, charge, life-time, spin etc.
- Mass: An elementary particle has always the same rest mass. If two particles have different rest masses, we consider them as different particles. The magnitude of the rest mass serves as the principal label which identifies the particles uniquely. The value of the rest mass may allow us to infer the existence of a particle even without a direct observation, just from the conservation of energy and momentum. The neutral pion π0 was discovered in this way.
- Charge: All known elementary particles have charge positive, negative or zero. Further, the charge is always conserved in any collision process.
- Average lifetime: All elementary particles, except photon, electron, proton and neutrinos are unstable and undergo decay into other elementary particle of smaller mass. The decay probability of a particular particle is, however, independent of the length of time the particle has lived. It is impossible to predict when a given (unstable) particle will decay. Hence an elementary (unstable) particle has an average lifetime, which is independent of the way the particle decays.
- Spin: Many elementary particles spin in a manner analogous to that of the earth on its axis, but with certain differences. The spin property forms a basis for the classification of elementary particles.
- Interactions: Four kinds of interactions between elementary particles are known: gravitational, weak, electromagnetic and strong. Their relative magnitudes are in the ratios 10-39: 10-13: 10-2: 1.